In Plain English: Tumor-Infiltrating Lymphocyte (“TIL”) Therapy for Advanced Melanoma

By Kim Margolin, M.D., FACP, FASCO
Note: We are rerunning an update on this TIL therapy article that originally ran in our newsletter and on our website in November of 2022 because of the February 2024 FDA accelerated approval of lifileucel (Amtagvi). Lifileucel is the cell therapy product, and the overall regimen is detailed below.
Background—Immunotherapy of Melanoma
The explosion of immunotherapy over the past decade has revolutionized the treatment of melanoma and nearly every other adult type of cancer. As a reminder, this form of treatment stimulates the body’s immune system to find, attack and kill tumor cells. Of all cancers, melanoma has benefited the most from immunotherapy, in large part because the mutations caused by extensive sun damage lead to changes in the genetic material of the malignant cells that can be recognized by cells of the immune system, which are further stimulated by a variety of forms of immunotherapy. Additionally, because melanoma is so resistant to chemotherapy and radiation, the relative benefits of immunotherapy are greater in patients with melanoma, who have few other choices for effective treatments.
What are TIL cells? What is TIL therapy?
TIL cells are tumor-infiltrating lymphocytes and are composed of CD8 and CD4 cells, the so-called killers and helpers of our immune system that are critically important in the immunologic fight against cancer as well as certain kinds of infections. TIL therapy refers to a treatment that involves extracting T lymphocytes from a patient’s tumor and growing them in special liquid containing lymphocyte growth factors. This liquid expands the numbers, enhances the activity of T lymphocytes, and leaves the tumor cells to die off. TIL cells are then given back to the patient via intravenous (in the vein through a needle) infusion. In successful treatments, these TIL cells will find, attack, and kill tumor cells. The factors that determine whether a patient will go into remission from TIL therapy are not well understood, but the number of cells that can be given may be an important factor, so the goal of laboratory expansion of TIL cells is usually several billion cells.
The TIL cell approach for advanced melanoma has been under investigation for nearly 35 years, starting in the mid-1980s in the laboratories at the National Cancer Institute’s Surgery Branch. There, Dr. Steve Rosenberg worked tirelessly and trained dozens of investigators (surgeons, medical oncologists, and laboratory scientists) to develop and improve on methods and outcomes of TIL therapy. While much of this research has been preclinical (in animals, where the conditions can be manipulated and the outcomes measured in much shorter timeframes than in people), an extensive series of clinical trials—testing the new therapy in people with cancer—has provided new insights. These insights include the best ways to obtain the tumor that the cells are taken from, cultivate the TIL cells, select the best cells to give back to the patient, and treat the patient with additional immune system-directed medications to optimize the anti-tumor effects of the TIL cell therapy.
Who can get TIL? How does the treatment work?
To date, the patients who have been considered likely to benefit from TIL therapy have had metastatic melanoma that has already been treated with standard therapies without achieving a long-term remission. This means nearly all patients have already received the small number of standard treatments currently available for advanced melanoma: both of the immune checkpoint blocking antibodies ipilimumab (Yervoy) and nivolumab (Opdivo), Opdualag (a relatively new combination of relatlimab, another immune checkpoint blocking antibody distinct from anti-CTLA-4 or anti-PD-1), plus nivolumab, or pembrolizumab (Keytruda) alone. If the patient’s tumor tested positive for the BRAF mutation, patients must also have had exposure to one of the three pairs of oral medications that target malignant cells with this mutation. Some patients have also received interleukin-2 (IL-2), the T cell growth and activation factor that is used in small amounts in laboratory preparation of TIL. In high doses, IL-2 causes T lymphocytes in the blood and tumor to multiply and become activated and provides a modest chance of remission when given alone to patients with melanoma. However, high doses of IL-2 can also cause a lot of (reversible) side effects and dangers to the patient. The TIL regimen also includes a few doses of high-dose IL-2, as detailed below, but IL-2 is stopped if there are dangerous effects, so this component of the regimen is not likely to pose serious risks or discomforts to melanoma patients undergoing TIL therapy. The FDA approval for lifileucel is restricted to patients who have already received one or more of the standard treatments noted above, including targeted therapy for those with a BRAF-mutated melanoma.
As shown in the diagram below, the TIL cell regimen begins with patient assessment, making sure that candidates for this therapy have a safely accessible tumor—one that can be partially or fully removed with a small surgical procedure. Patients with tumors in the brain, the spinal cord, bones, and other less safely accessible locations would not qualify for TIL therapy. Patients must also be in generally good medical and physical condition, which means they are judged by their doctor to be healthy enough to get through all phases of the regimen, including several days of chemotherapy and a one to two-week period of low blood counts that necessitate supportive care with antibiotics, blood transfusions, and potentially intravenous feeding.
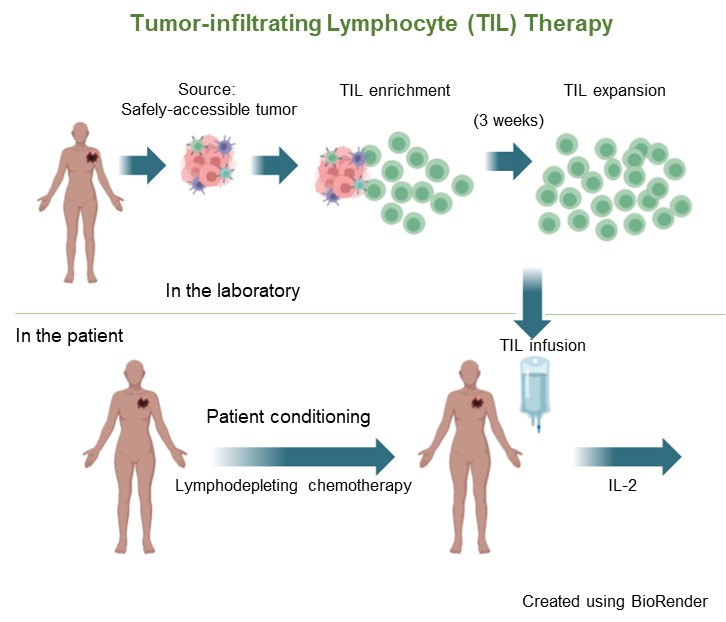
The next step after tumor excision is chopping up the tissue into small fragments, placing them in an incubator at body temperature in growth medium for up to three weeks, and checking the cells for viability (must be alive) and freedom from contamination. Meanwhile, the patient is first prepared by the administration of two chemotherapy drugs over five to seven days. These drugs do not kill melanoma cells but lower the patient’s blood lymphocyte count dramatically (along with other cells originating in the bone marrow, which causes all blood cells to be reduced). The body’s reaction to this lymphocyte reduction (termed “lymphodepletion”) is to produce growth factors that stimulate its production of new lymphocytes. The infused TIL cells are also further expanded and stimulated. It is likely that in the near future, we will have purified and safely administered forms of these lymphocyte growth factors that can be given instead of chemotherapy and possibly also replace the IL-2 that is given after TIL infusion.
Because of the risks involved with very low blood counts for one to two weeks, patients must be hospitalized for the supportive measures detailed above and for close nursing and physician supervision during this time and during the two days of high-dose IL-2 that follow TIL infusion. Thereafter, patients are discharged from the hospital and monitored in the clinic.
What are the most current results of TIL cell therapy?
Most of the published results of TIL cell therapy for melanoma have come from the National Cancer Institute Surgery Branch mentioned above; additional data are from a small number of cancer centers, mostly in the U.S but also in other countries, where different research approaches have been taken to the optimization of this and related forms of cell therapy for cancer. On the average, about 1/3 of patients receiving TIL therapy go into remission. However, many of those patients still relapse subsequently, so it is clear that we need to find better approaches and better ways to select the best regimen and its timing for each patient.
The first trial ever to randomize TIL therapy against standard immunotherapy for advanced melanoma patients in first relapse following single-agent PD-1 blockade was reported by Haanen et al. at the 2022 annual meeting of the European Society of Medical Oncology. In this study, 168 patients with similar characteristics whose melanoma was not controlled with PD-1 antibodies were randomly assigned to receive either ipilimumab or undergo TIL therapy. The results strongly favored TIL therapy, with over twice (49%) the response rate of ipilimumab (21%). Average time without relapse or death were also over twice as long in patients who received TIL therapy. Of note is that patients who were randomized to ipilimumab did not have the opportunity to “cross over” to receive TIL therapy if they relapsed, so it is not possible to know from this study whether early use of TIL is better than later use of TIL. Now that we also have data showing about a 30% rate of remission for patients in relapse after single-agent PD-1 blockade with pembrolizumab or nivolumab who go on to receive ipilimumab plus nivolumab, we need to design trials to demonstrate whether TIL therapy is truly superior to other available options and discover the optimal timing. Perhaps all or nearly all patients with advanced melanoma should have a TIL product harvested from a resected tumor metastasis and stored frozen in a laboratory to be used later if the patient relapses following non-TIL therapy.
These exciting results for TIL cell therapy as an option and a hope for patients who do not achieve long term benefit from standard immunotherapy supported the February, 2024 accelerated FDA approval, which is based on smaller patient numbers than full approval, does not require randomization, and is judged to fulfill a critical unmet need. But it will still be necessary to perform the post-marketing, randomized trial needed for full FDA approval of TIL therapy for advanced melanoma. Further studies are ongoing to improve TIL therapy with other technical adjustments, patient selection, and optimization of the timing and sequence, as we have covered in other issues of In Plain English.
There remain many questions and potential obstacles to the approved and now-available form of TIL therapy for advanced melanoma, including cost, time requirements, and access issues. The practical ones concern the exorbitant cost of cell therapy—but considering that cell therapy is a “one and done” strategy, it may not prove to be more expensive than sequences of less complex treatments punctuated by relapses. The length of time required to prepare the TIL cell product has also been reduced as laboratory advances are made—currently to only about three weeks. Access to centers with experience that can safely administer this therapy may be challenging, but many of the principles follow those of other forms of high-dose therapy, like bone marrow transplantation (but without the need for a donor), which is now widely available, safe and effective.
Taken together, the pooled results of TIL cell therapy from a number of U.S. and ex-U.S. centers as well as the industry-sponsored studies are remarkably good, considering that these studies most often enrolled patients who had exhausted all other therapies with known benefit. But we must not lose sight of the fact that the rising tide also lifts all boats, so that while TIL cell and related strategies are being worked on to improve their outcomes, safety, lower costs and enhance accessibility, other treatments for melanoma and other cancers are also in the works. Competition is good, as it brings out the creativity of scientists and the enthusiasm of clinical investigators. It is possible that sometime in the near or distant future, TIL cell therapy will have become not only commonplace but maybe even obsolete, moving aside in favor of treatments that are even more effective, safe, well-tolerated and affordable. Look for those advances in coming issues of In Plain English.
Questions on this article may be submitted to Alicia@AIMatMelanoma.org

Dr. Margolin is a Medical Director of the SJCI Melanoma Program, St. John’s Cancer Institute. She worked at City of Hope for 30 years and also held faculty positions at the Seattle Cancer Care Alliance/University of Washington and at Stanford University. Among her academic achievements were long-term leadership of the Cytokine Working Group, leadership involvement in the Cancer Immunotherapy Trials Network, participation in the Southwest Oncology Group’s Melanoma Committee, and many positions in the American Society of Clinical Oncology and the Society for Immunotherapy of Cancer. Dr. Margolin has reviewed grants for many cancer-related nonprofit organizations and governmental agencies. She has also served as a member of the Oncology Drugs Advisory Committee to the FDA, the American Board of Internal Medicine’s Medical Oncology certification committee, and the Scientific Advisory Committee of the European Organization for the Research and Treatment of Cancer.
Dr. Margolin collaborates with AIM at Melanoma to write our In Plain English articles to provide timely updates on new developments for patients, caregivers, and other individuals with an interest in medical advances in melanoma.
Recent Posts

Webinar: Post-ASCO Melanoma Research Update

Melanoma Monday: Every Day. Everywhere.

Lilly Pulitzer and AIM at Melanoma Partner for 5th Annual Community Giving Event

KIMMTRAK doubles the likelihood of being alive at five years for firstline HLA-A*02:01+ patients with metastatic uveal melanoma


