Subcutaneous Immunotherapies for Melanoma: What You Need to Know

What Are Subcutaneous Melanoma Immunotherapies and How Do They Differ From Intravenous Therapies?
Immunotherapies—therapies that use the immune system to target and destroy cancer cells—have been used to treat melanoma since 2011, and include Yervoy (ipilimumab), Keytruda (pembrolizumab), Opdivo (nivolumab), and Opdualag (relatlimab combined with nivolumab).
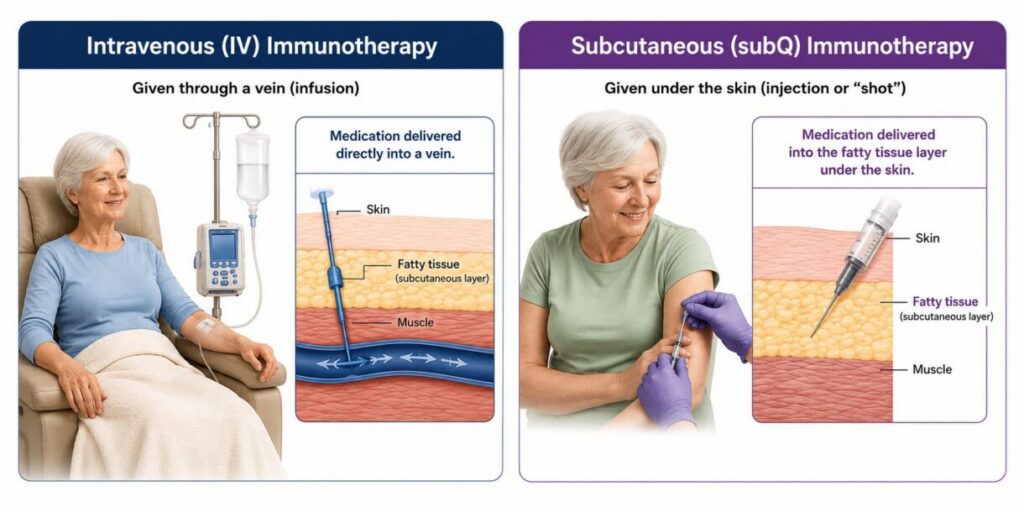
Until recently, these immunotherapies could only be administered intravenously (in a vein)—often referred to as an “IV.” If you receive your immunotherapy intravenously, you will sit in a chair while a healthcare provider inserts a needle into a vein, usually in your arm, and the medication will flow through a tube directly into your vein. This process is also known as infusion. Because the medication is delivered through a tube into your vein, it flows at only a modest speed. The whole process can take 30-60 minutes or longer. Infusion generally must occur in a cancer center or hospital outpatient setting.
Over the past few years, the FDA has approved subcutaneous formulas of two melanoma immunotherapies, specifically Keytruda and Opdivo. Subcutaneous means they are administered in the fatty tissue layer between the skin and muscle—or, in everyday language, it means delivery via an injection, or a “shot.” The medication is the same immune triggering therapy received during infusion (Keytruda or Opdivo), along with an enzyme that helps the delivery and distribution of the immunotherapy in your body, since it’s not going directly into a vein. This subcutaneous delivery method, often called subQ for short, is easier to administer, takes less time, and doesn’t require the outpatient setup that infusion requires.
The two FDA approved subQ immunotherapies for melanoma are called Opdivo Qvantig and Keytruda Qlex.1,2
Are Subcutaneous Immunotherapies as Effective as their Infusion Counterparts?
The most important information to know is that for these two melanoma treatments, the subQ and infusion versions are the same cancer medication, delivered differently. So, whether you receive Opdivo Qvantig as a subQ injection or Opdivo as an infusion, you are receiving the same cancer medication. The same is true for Keytruda Qlex and Keytruda.
Not surprisingly then, in clinical studies, Opdivo Qvantig was shown to have similar and consistent efficacy (effectiveness) compared to intravenous Opdivo. Likewise, there were no notable differences in efficacy for patients who received Keytruda Qlex compared to patients who received intravenous Keytruda.1,2
How do Subcutaneous Immunotherapies Work?
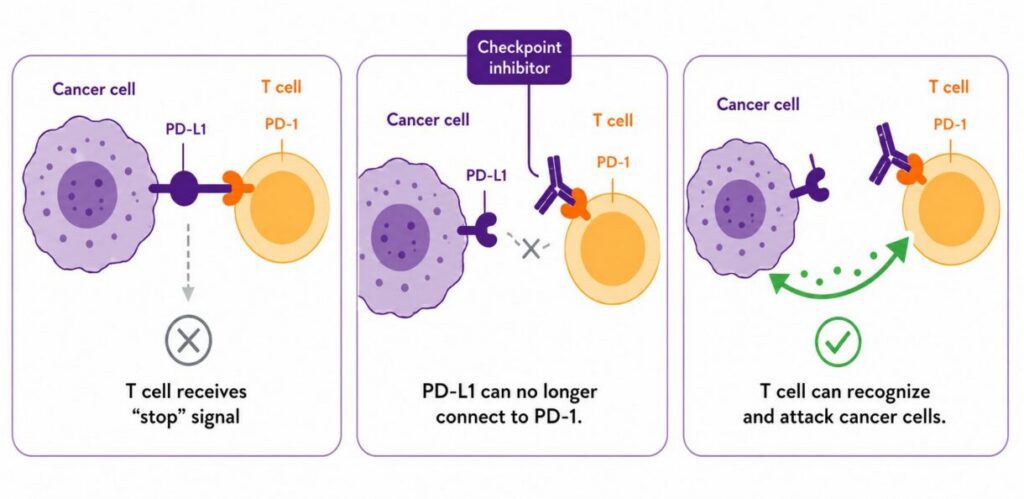
All immunotherapies—Keytruda and Keytruda Qlex and Opdivo and Opdivo Qvantig—work by increasing the ability of the body’s immune system to find and kill cancer cells. Cancer is tricky, and it is often able to shut down our immune system’s ability to recognize it as a threat. These drugs reengage and reinvigorate our immune system so that it can recognize and destroy melanoma cells.
Specifically, these drugs are designed to bind to a cellular target called PD-1, which blocks the interaction between PD-1 and its ligands, PD-L1 and PD-L2. Once the interaction is blocked, the immune system is more able to recognize the tumor cells as something it should destroy. You’ll often hear these immunotherapies—whether delivered via subQ or infusion—referred to as “anti-PD-1s.”
Opdivo Qvantig is a combination of the immunotherapy drug nivolumab and an enzyme called hyaluronidase. The addition of hyaluronidase increases the distribution of nivolumab throughout the body and the absorption of nivolumab into the tissues, which is necessary because the medication is not going directly into your circulatory system as it does when it is infused.2
Keytruda Qlex is a combination of the immunotherapy drug pembrolizumab and berahyaluronidase. Berahyaluronidase is an enzyme that breaks down hyaluronic acid in the skin which allows pembrolizumab to be better absorbed under the skin. Because the medication is not being infused into your circulatory system, this enzyme is necessary.1
Why Were Subcutaneous Cancer Immunotherapies Developed?
Research often seeks new and better treatments, but sometimes research yields developments that offer a variation of current treatments. Such is the case with subQ melanoma treatments.

Infusion generally must occur in a cancer center or hospital outpatient setting and takes 30 or more minutes to administer, which can be burdensome to both patients and the healthcare system. By contrast, subQ therapies may be administered in an office setting or clinic, and it takes a fraction of the time to administer.
For patients who do not live near a cancer center or hospital, subcutaneous injections can save travel time to and from the cancer center. Even if you do live close to a cancer center, subcutaneous injections can save time for you because the medication is easier to administer and takes less time to do so. For healthcare providers, the ability to treat a patient in a fraction of the time and in the same room as a routine appointment is also beneficial.
Subcutaneous Injections and Infusion Administration Compared
Subcutaneous
| If you and your healthcare provider opt for intravenous administration of your melanoma immunotherapy, you’ll be treated in a cancer center or hospital outpatient setting. You’ll receive your infusions as you sit in a chair in what is often called an infusion center. Intravenous infusion delivers the medicine directly into your circulatory system and is therefore quickly distributed within your body. A healthcare provider will first clean your skin and insert a needle into a vein most often at the inside joint of your forearm. Then, a tube will be attached so that the medication can flow through the tube and into your vein. You are monitored by the healthcare staff to make sure you don’t have any type of reaction. The infusion itself takes 30 minutes, and the whole process can take an hour or more, depending on the time it takes to set up your IV; to receive the medication from the pharmacy once your IV is prepped; and to monitor you for a reaction. |
Infusion
| If you and your healthcare provider opt for subQ administration of your melanoma immunotherapy, you may receive your injection at a healthcare provider’s office or clinic. Since it does not need to be administered at a cancer center or similar location, there are likely multiple options for administration location. Administration may be in a somewhat private area as the injection is given in your stomach area or thigh. A healthcare provider will first clean your skin at the injection site. Then you will be injected, which will take one to five minutes. You will be watched to make sure you have no reaction to the injection. While the injection itself takes only a few minutes, the overall treatment time takes longer and will include preparation of the injection, the injection itself, and the time to watch for a reaction. Keytruda Qlex can be given via an injection of one minute duration if administered every three weeks or an injection of two minutes duration if administered every six weeks.1 Opdivo Qvantig is given via an injection of three to five minutes’ duration every two, three, or four weeks, depending on the dose you receive.2 |
Are the Safety and Side Effects of Subcutaneous Immunotherapies the Same as their Infusion Formulations?
As noted previously, the subQ and infusion versions of these two melanoma treatments are the same cancer medication, delivered differently. So, whether you receive Keytruda Qlex as a subQ or Keytruda as an infusion, you are receiving the same cancer fighting medication. The same is true for Opdivo Qvantig and Opdivo.
Not surprisingly, the results from clinical studies with Opdivo Qvantig showed that the safety profile is similar to that for intravenous Opdivo. Also not surprisingly, Keytruda Qlex and intravenous Keytruda have comparable safety.1,2
With subcutaneous delivery, injection site reactions can occur and involve redness, itching, and swelling. These complications can be managed with a cold compress or with over-the-counter pain medications. With infusion delivery, pain at the IV site can occur and this can be managed similarly. It is also possible for an IV site to get infected.
Are Patients Monitored the Same Way on Subcutaneous Injection vs Infusion?
In both cases—subQ and infusion delivery—patients are monitored while they are receiving the medication. Again, the safety profiles for both Keytruda Qlex/Keytruda and Opdivo Qvantig/Opdivo are comparable.1,2
Who is Eligible for Subcutaneous Melanoma Immunotherapy?
SubQ immunotherapies were relatively recently approved by the FDA (Opdivo Qvantig in December 2024; Keytruda Qlex in September 2025). If you are interested in receiving your treatment via subQ injection, you should ask your healthcare provider whether it’s the right choice for you.
- Keytruda Qlex is approved for the treatment of adult patients with unresectable (cannot be removed with surgery) or metastatic (Stage IV) melanoma. Keytruda Qlex is also approved for the adjuvant treatment of adult and pediatric patients 12 years and older with Stage IIB, IIC, or III melanoma following complete resection (removal by surgery).1
- Opdivo Qvantig is approved for adult patients with unresectable melanoma or metastatic melanoma; for adult patients with unresectable or metastatic melanoma following combination treatment with intravenous nivolumab and ipilimumab; and for the adjuvant treatment of adult patients with completely resected Stage IIB, Stage IIC, Stage III, or Stage IV melanoma.2
Which Immunotherapy Formulation May Be Right for You?
Results from studies (in various cancers) indicate that patients tend to prefer subcutaneous therapies and that they experience greater satisfaction with them compared to intravenous therapies.3
It is important to discuss with your healthcare provider whether subQ or infusion is best for you. It’s also important to consult with your insurance provider on coverage for either treatment.
Reasons you and your healthcare provider might choose subcutaneous injections: 3-9
Time
- Less time is needed for preparation and administration of the treatment, so you spend less time receiving treatment
- SubQ may be available at a location physically closer to you than a cancer center or hospital, so you may spend less time traveling to and from treatment appointments
Complications and Preference
- Subcutaneous injections are beneficial if you experience anxiety or pain during IV infusions or if you have fragile veins or veins that are difficult to access
- There is no need for venous access, so there is no need to repeatedly puncture your blood vessels
- There may be less pain at the injection site because the medication does not go into your vein
- There may be fewer complications such as infection that can occur with IV therapies
- There is a choice of injection site, in the thigh or abdomen, which provides flexibility
Reasons you and your healthcare provider might not choose subcutaneous injections:
Time
• If you also receive other IV treatments it may not make sense to receive an additional treatment in a different way, as that would add more time to complete your treatment regimen
• If you have other reasons to travel to the cancer center, such as for lab work or healthcare provider appointments, you can combine visits with infusion
Complications and Preference
- You may prefer IV administration if you experience anxiety about shots
- IV administration might be preferable if you are uncomfortable with the possibility of unintended injection into a muscle
- You may prefer IV administration if the location of the injection (stomach area or thigh) is not comfortable for you
- IV administration might be preferable if you are uncomfortable with the possibility of redness, itching, and swelling at the injection site
Change
- If you started your therapy as an infusion, your healthcare provider may prefer to keep you on infusion
To learn more about FDA-approved immunotherapies and other approved treatments for melanoma, visit the AIM at Melanoma FDA Approved Drugs resource: https://www.aimatmelanoma.org/fda-approved-drugs/
References

